MonaLisa Touch
As for all new patients to this specialist practice (see First Visit) a referral from your doctor is required.
Please note that this website provides a guide and general information only. Not everyone is suitable for this treatment, and if you are interested then Doctor Burrows will assess your clinical scenario and advise you on what your next steps might be.
Treatment for a Dry or Itchy Vagina
MonaLisa Touch is a treatment for women who have a dry and itchy vagina. It is a non-invasive alternative to Hormone Replacement Therapy for the treatment of symptoms related to Vaginal Atrophy.
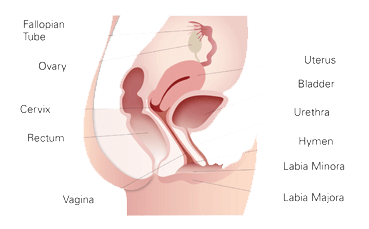
What is Vaginal Atrophy?
Vaginal Atrophy occurs as women age. Common symptoms can include: dyspareunia, urinary incontinence, itchiness, burning, dryness, mild prolapse and laxity, pelvic pain, pain with intercourse, urinary incontinence or recurrent urinary infection.
How will I benefit from the treatment?
Most patients experience a noticeable improvement after just one MonaLisa Touch treatment however a course of three treatments month apart is recommended. Patients can return to their normal daily activities immediately following a MonaLisa Touch procedure. The post treatment instructions are to avoid any sexual activity and avoid strenuous exercise for 5 days.
What is involved?
It is a five minute, pain free procedure that has no downtime and is suitable for almost all patients suffering from the effects of Vaginal Atrophy. MonaLisa Touch is a low risk procedure that provides longer lasting improvements than HRT. However, it can be used in conjunction with HRT, as an alternative treatment for patients who are not suitable for HRT or for those patients who have found HRT to be ineffective. Dr Burrows will discuss your particular symptoms prior to the procedure and allow time to answer any questions or concerns.
How does it work?
MonaLisa Touch works by stimulating the body’s own regenerative processes to create more hydrated and healthy cells and to improve the vascularity of the vaginal mucosa. This has a direct effect on the integrity and elasticity of the vaginal wall and increases the acidity of the vaginal environment to more normal levels. In general terms MonaLisa Touch improves the function and health of the vagina.
Recent Developments
-
 Click here to find out about The Latest Clinical Data on Monalisa Fractional CO2 Laser
Click here to find out about The Latest Clinical Data on Monalisa Fractional CO2 Laser -
 Click here to find out about Monalisa touch brochure
Click here to find out about Monalisa touch brochure
You will need the Adobe Reader to view and print the above documents. 





